Home Tags Posts tagged with "telomer"
telomer
According to US researchers, the Mediterranean diet offers a long life because it appears to keep people genetically younger.
Mediterranean diet’s mix of vegetables, olive oil, fresh fish and fruits may stop our DNA code from scrambling as we age, according to a study in the British Medical Journal.
Nurses who adhered to the diet had fewer signs of ageing in their cells.
The researchers from Boston followed the health of nearly 5,000 nurses over more than a decade.
The Mediterranean diet has been repeatedly linked to health gains, such as cutting the risk of heart disease.
Although it’s not clear exactly what makes it so good, the Mediterranean diet’s key components – an abundance of fresh fruit and vegetables as well as poultry and fish, rather than lots of red meat, butter and animal fats – all have well documented beneficial effects on the body.
Foods rich in vitamins appear to provide a buffer against stress and damage of tissues and cells. And it appears from this latest study that a Mediterranean diet helps protect our DNA.
The researchers looked at tiny structures called telomeres that safeguard the ends of our chromosomes, which store our DNA code.
These protective caps prevent the loss of genetic information during cell division.
As we age and our cells divide, our telomeres get shorter – their structural integrity weakens, which can tell cells to stop dividing and die.
Experts believe telomere length offers a window on cellular ageing.
Shorter telomeres have been linked with a broad range of age-related diseases, including heart disease, and a variety of cancers.
In the study, nurses who largely stuck to eating a Mediterranean diet had longer, healthier telomeres.
No individual dietary component shone out as best, which the researchers say highlights the importance of having a well-rounded diet.
Independent experts said the findings were interesting but by no means conclusive.
[youtube zbUzSotKUUc 650]
Scientists say life expectancy is written into our DNA and could be seen from the day we are born.
They have found a way to predict how long someone will live – by measuring their genes as a baby
It all depends on the length of the telomeres, which are described as “acting like the plastic ends on shoelaces” to protect chromosomes from wear and tear.
Telomeres are being studied extensively – and are thought to hold the key to ageing.
The longer your telomeres, the longer you will live – dependent, of course, on not dying accidentally, from disease or from lifestyle factors.
It was known they could be shortened by life choices, including smoking and stress. But this is the first indication that our lifespan might be predetermined from birth.
In the future, tests may allow people to know their expected lifespan from a very early age – if they want to.
Professor Pat Monaghan, who led the Glasgow University study, said: “The results of this research show that what happens in our bodies in early life is very important.
“It is not understood why there are variations of telomere length but if you had a choice, you would want to be born with longer telomeres.
“If you were to test this, I don’t think anyone would want to know – it would just make you miserable. But it must be remembered that how you live has a big effect. This isn’t quite a case of nature overtaking nurture.”
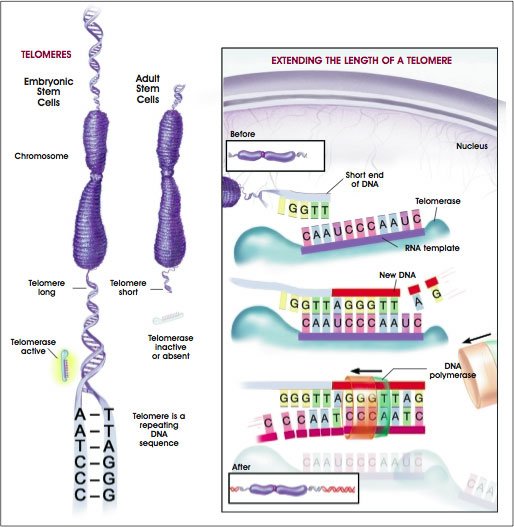
Telomeres are important because they stop DNA from unraveling, but they begin shortening from the moment we are conceived
The study – which used zebra finches, one of Australia’s most common bird species – is the first to measure telomere lengths at regular intervals through an entire life. With people, it is usually only the elderly who are studied because of the timescales involved.
Blood cell samples were taken from 99 finches, starting when they were 25 days old.
The results exceeded even the researchers’ expectations. The birds with the shortest telomeres did tend to die first – from as early as seven months after the start of the trial.
But one bird in the group with the longest telomeres survived to almost nine years old.
Professor Monaghan said: “These birds were dying of natural causes. There were no predators, no diseases and no accidental deaths. This was showing their capacity for long life.”
The results hold huge implications for humans, whose telomeres work in the same way.
Telomeres are important because they stop DNA from unraveling, but they begin shortening from the moment we are conceived.
The longer they are, the better for an individual because when they get too short, they stop working.
DNA is then no longer protected and errors begin to creep in when cells divide. When this happens – usually in middle age – the skin begins to sag and the immune system becomes less efficient. Faulty cells also lead to a growing risk of conditions such as diabetes and heart disease.
The university’s institute of biodiversity, animal health and comparative medicine has published its groundbreaking research in the Proceedings of the National Academy of Sciences USA.
In the next stage of their research, the Glasgow scientists will look at what causes telomeres to shorten – including inherited and environmental factors – to make it possible to predict life expectancy more accurately.
A new drug derived from autumn crocus extracts could wipe out tumors in a single treatment with minimal side effects, according to University of Bradford researchers in UK.
The British researchers from University of Bradford have turned a chemical found in crocuses into a “smart bomb” that targets cancerous tumors.
The most important fact is that healthy tissue is unharmed, reducing the odds of debilitating side effects.
Unlike other side effect-free products, the new drug is able to kill off more than one type of the disease, including breast cancer, prostate cancer, lung and bowel cancer.

The miraculous drug is based on colchicine, an extract from the autumn crocus (Colchicum autumnale), known also as meadow saffran
Potentially, all solid tumors could be vulnerable to drugs developed this way, meaning it could be used against all but blood cancers.
In some tests of the drug, half of tumors vanished completely after a single injection and the results will be presented at the British Science Festival this week.
The miraculous drug is based on colchicine, an extract from the autumn crocus (Colchicum autumnale), known also as meadow saffran, and is at an early stage of development, being so far tested only on mice.
But the University of Bradford researchers are optimistic about drug’s potential in humans.
Professor Laurence Patterson said:
“What we have designed is effectively a <<smart bomb>> that can be triggered directly at any solid tumour without appearing to harm healthy tissue.
“If all goes well, we would hope to see these drugs used as part of a combination of therapies to treat and manage cancer.”
Colchicine has long been known to have anti-cancer properties but has been considered too toxic for use in the human body.
To solve this issue, the specialists attached a chemical “tail” to the substance, deactivating it until it reaches the cancer.
Once colchicine reaches the target, the tail is cut off by an enzyme called MMP (Matrix metalloproteinase involved in cell proliferation), which is found in tumors.
Once the tail is removed, the drug is activated, which then attacks and breaks down the blood vessels supplying the tumors with oxygen and nutrients.
Cancerous tumors use the blood supply to spread around the body and it is hoped that the treatment, called ICT2588, will also combat this.
The first tests on humans could start in as little as 18 months.
If the treatment will be successful, the drug could be on the market in 6 to 7 years.
Medical scientists have found a new form of mechanism leading to hereditary cancer susceptibility, demonstrating that minor changes in certain anti-cancer genes might work as magnets to draw altering “biochemical tags”.
The tags successfully switch these genes off as well as predispose families to a higher risk associated with the condition. The researchers, from the University of New South Wales (UNSW), consider that a minor spelling error affecting just one letter in the DNA sequence close to the beginning of the gene is actually what draws in the biochemical marking, named methylation. This methylation switches genes off, thus has specific influences on the DNA.
“Methylation sits on top of our DNA, and provides the instructions to turn the gene off,” pointed out Dr Megan Hitchins.
In one well studied cause of hereditary cancer, alterations in the cancer-prevention gene MLH1 are transferred from mother or father to children. This situation generates up to 80 percent risk of developing bowel cancer, uterine cancer and other malignancies.
On the other hand, a number of family members with hereditary cancer do not have spelling mistakes in MLH1, but alternatively possess methylation placed on the gene.
“When the methylation attaches to the MLH1 gene in these families, it causes it to be completely switched off and as a consequence cancer develops,” affirmed head of the adult cancer program at the Lowy Cancer Research Centre, Professor Robyn Ward. “But until now, we did not understand how these methylation tags were being passed from parent to child.”

Hereditary cancer study: certain anti-cancer genes act like a magnet for methylation that disappears in semen or ova and reappears in child
In the study the researchers investigated three generations of a large family, who had experienced cancer at young age, but in whom no spelling mistakes characteristic for this kind of inherited cancer syndrome had been identified. Noticeably a number of family members from all generations had methylation tags on their gene.
“In this family, biochemical tags attached to the MLH1 gene were present in all three generations. This was intriguing since these markers are usually removed during the production of eggs and sperm. What we found was that a subtle change near the gene was acting like a magnet to attract methylation. So it was not the methylation itself that was being passed from parent to child, but rather the DNA change, and this acted as a methyl magnet,” Dr Megan Hitchins said.
The methylation had disappeared in the semen and ova and after that regenerated in every brand-new generation, the researchers explained.
Professor Robyn Ward said the study had specified the trigger of cancer in this family and it had provided new options for genetic diagnosis, counselling as well as early treatment in other families vulnerable to hereditary cancer.
The group is additionally studying the utilization of specific medications (Epigenetic Reversal Drugs) to remove the methylation in cancer, thus to switch the anti-cancer genes back on again. Later on these medicines might be administered in order to create a more specific method of cancer therapy and perhaps prevention.
Additional scientists linked to this research have been the Genetic Services of Western Australia and School of Paediatrics and Child Health, University of Western Australia.
The study on hereditary cancer was published in the primary international journal “Cancer Cell”.
On Wednesday, August 17, the Food and Drug Administration approved Zelboraf, a drug for BRAF V600E mutation-positive metastatic melanoma. Along with skin cancer drug, FDA approved Companion Diagnostic, a mutation diagnostic test.
“Today’s approval of Zelboraf and the (companion) test is a great example of how companion diagnostics can be developed and used to ensure patients are exposed to highly effective, more personalized therapies in a safe manner,” said Alberto Gutierrez, head of the FDA’s Office of In Vitro Diagnostic Device Evaluation and Safety.

Zelboraf may help metastatic melanoma patient to live longer
Zelboraf and its Companion Diagnostic are co-developed by Roche (Genentech unit) and Plexxikon, Daiichi Sankyo Group member. Roche and Daiichii will promote this medicine in United States. Zelboraf will be available within two weeks and will be distributed through specialty pharmacies (mail-order pharmacies).
Zelboraf (vemurafenib) is a targeted medicine for inoperable or metastatic melanoma BRAF V600E mutation-positive.
“As the first personalized medicine approved for the treatment of patients with BRAFV600E mutation-positive melanoma, Zelboraf represents a significant new treatment option for these patients suffering from inoperable or metastatic melanoma. We are grateful for the clinical trial participants, clinical investigators, collaborators and dedicated employees, who have all contributed to this important advancement in cancer treatment,” said K. Peter Hirth, Ph.D., chief executive officer of Plexxikon.
The cobas 4800 BRAF V600 Mutation Test identifies tumors that carry the BRAF V600E mutation. In this way the doctors will know what person with inoperable or metastatic melanoma can be treated with Zelboraf. This personalized drug has no known beneficial effect on other skin cancers, thus it is important to have a diagnostic test before taking Zelboraf pills.
“The cobas BRAF Mutation Test has improved sensitivity, accuracy and speed compared to other commonly used, unapproved detection methods,” said Paul Brown, head of Roche Molecular Systems.
Studies have shown that Zelboraf increases the survival rate in patients with BRAF V600E mutation-positive melanoma.
According to the American Cancer Society, melanoma, a type of skin cancer, is one of the deadliest cancers, with a five-year survival rate of 15 percent for people with advanced melanoma. It is estimated that BRAF mutations appear in about half of all cases of melanoma and may lead to uncontrolled cell growth.
More information about ongoing Zelboraf studies is available at www.roche-trials.com or www.clinicaltrialsregister.eu (in the EU) or www.clinicaltrials.gov (in the US).
Zelboraf might have side effects. The patients should talk to their doctors about their conditions prior the medication and carefully observe the changes that might appear during the treatment.
“Patients should call their doctor for medical advice about any side effects. Patients or their caregivers are encouraged to report negative side effects of prescription drugs to the FDA at 1-800-FDA-1088 or at www.fda.gov/medwatch. They may also report side effects to Genentech at 1-888-835-2555.
Patients should read the Zelboraf full Prescribing Information and Medication Guide for additional important safety information at www.zelboraf.com.”
In March, the FDA approved Bristol-Myers Squibb’s Yervoy (ipilimumab), an intravenous medicine for patient with advanced melanoma. Roche and Bristol have agreed to collaborate in order to find out if these two medicines are safe and effective if taken together. Roche submitted drug applications for Zelboraf in the EU, Switzerland, Australia, New Zealand, Brazil, India, Mexico and Canada.
[googlead tip=”patrat_mediu” aliniat=”stanga”]A new treatment for leukemia had amazing results, surprising even the researchers who designed it. The new treatment has eradicated the cancer cells present in the first three patients tested bodies.
Early results of a clinical trial showed that genetically engineered T cells eradicate leukemia cells and thrive.
Scientists from the University of Pennsylvania have genetically engineered patients’ T cells — a type of white blood cell — to attack cancer cells in advanced cases of a common type of leukemia.
The first two of three patients studied, who received the innovative treatment, have been cancer-free for more than one year. In the case of the third patient, over 70% of cancer cells were removed, according to the researchers.
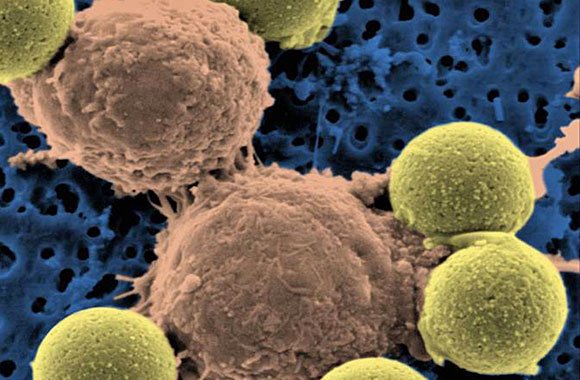
"Microscopic image showing two T cells binding to beads, depicted in yellow, that cause the cells to divide. After the beads are removed, the T cells are infused into cancer patients." (Dr. Carl June / Pennsylvania Medicine)
“In just three weeks, tumors were destroyed, the effect being more violent than we ever have imagined,” said Dr. Carl June, one of the researchers involved in the study.
“Each cell can destroyed thousands of cancer cells,” said June, “each patient have been removed tumors from at least 900 grams.”
“A huge accomplishment”
[googlead tip=”vertical_mare” aliniat=”dreapta”] “This is a huge accomplishment — huge,” said Dr. Lee M. Nadler, dean for clinical and translational research at Harvard Medical School, who discovered the molecule on cancer cells that the Pennsylvania team’s engineered T cells target.
Innovative treatment is using patients’ own T cells, which are extracted from body cells and then genetically modified to attack cancer cells and to multiply and then reintroduced into patients’ blood.
Findings of the trial were reported Wednesday in the New England Journal of Medicine and Science Translational Medicine.
According to LA Times report, for building the cancer-attacking cells, the researchers modified a virus to carry instructions for making a molecule that binds with leukemia cells and directs T cells to kill them. Then they drew blood from three patients who suffered from chronic lymphocytic leukemia and infected their T cells with the virus.
When they infused the blood back into the patients, the engineered T cells successfully eradicated cancer cells, multiplied to more than 1,000 times in number and survived for months. They even produced dormant “memory” T cells that might spring back to life if the cancer was to return.
On average, the team calculated, each engineered T cell eradicated at least 1,000 cancer cells.
Side effects included loss of normal B cells, another type of white blood cell, which are also attacked by the modified T cells, and tumor lysis syndrome, a complication caused by the breakdown of cancer cells.
“We knew [the therapy] could be very potent,” said Dr. David Porter, director of the blood and marrow transplantation program at the Hospital of the University of Pennsylvania in Philadelphia and a coauthor of both papers, which were published in the New England Journal of Medicine and Science Translational Medicine.
“But I don’t think we expected it to be this dramatic on this go-around.”
Bone marrow transplants from healthy donors have been effective in fighting some cancers, including chronic lymphocytic leukemia, but the treatment can cause side effects such as infections, liver and lung damage, even death.
“1/5 of bone marrow transplant recipients may die of complications unrelated to their cancer,” Porter said.
Researchers have been working for many years to develop cancer treatments that leverage a patient’s immune system to kill tumors with much greater precision.
Specialists not involved in the trial said the new discovery is very important because it suggested that T cells could be adapted to destroy a range of cancer cells, including ones of the blood, breast or colon
“It is kind of a holy grail,” said Dr. Gary Schiller, a researcher from UCLA’s Jonsson Comprehensive Cancer Center who was not involved in the trial.
“It would be great if this could be applied to acute leukemia, where there is a terrible unmet medical need,” UCLA’s Schiller said.
Dr. David Porter added:
“Previously efforts to replace risky bone marrow transplants with such engineered T cells proved disappointing because the cells were unable to multiply or survive in patients.”
“This time, the T cells were more robust because the team added extra instructions to their virus to help the T cells multiply, survive and attack more aggressively.”
“About 15,000 patients are diagnosed with chronic lymphocytic leukemia every year. Many can live with the disease for years. Bone marrow transplants are the only treatment that eradicates the cancer.”
[googlead tip=”lista_mare” aliniat=”stanga”]Dr. David Porter cautioned that these were preliminary results and the scientists plan to continue the trial, treating more patients and following them over longer periods.
“The researchers also would like to expand the work to other tumor types and diseases,” Porter said.
The hope, scientists said, is that the method would work for cancers that can kill more ruthlessly and rapidly.
[googlead tip=”patrat_mare” aliniat=”stanga”]
Progressive telomere shortening characterizes familial breast cancer patients
Telomere chromosome
Telomeres, the complex structures that protect the end of chromosomes, of peripheral blood cells are significantly shorter in patients with familial breast cancer than in the general population. Results of the study carried out by the Human Genetics Group of the Spanish National Cancer Research Centre (CNIO), led by Javier Benitez, to be published in open-access journal PLoS Genetics on July 28th, reflect that familial, but not sporadic, breast cancer cases are characterized by shorter telomeres. Importantly, they also provide evidence for telomere shortening as a mechanism of genetic anticipation, the successively earlier onset of cancer down generations.
Mutations in two DNA repair genes, BRCA1 and BRCA2, characterize some, but not all, instances of hereditary breast cancer. Non-BRCA1/2 breast cancer families are heterogeneous, suggesting the existence of other genes conferring susceptibility. The group has investigated the role of telomere length in hereditary breast cancer based on previous information suggesting, first, that short telomeres and subsequent genomic instability contribute to malignant transformation; second, that genetic anticipation occurs in breast cancer families and, third, that telomere shortening is associated with anticipation in other genetic diseases. [googlead tip=”vertical_mare” aliniat=”stanga”]
By analyzing telomere length differences between mothers and daughters from breast cancer families, the authors demonstrated that genetic anticipation is associated with a decrease in telomere length in affected daughters relative to their mothers.
The results allowed the authors not only to conclude that women carrying BRCA1/2 mutation have chromosomes with short telomeres, but also to describe for the first time that genetic anticipation in breast cancer could be explained by telomere shortening. In addition, the study expands the field of research concerning genetic predisposition to breast cancer to include genes involved in telomere maintenance. The significance of generational changes in telomere length has interesting potential clinical applications in the management of familial breast cancer, and could be extended to other hereditary cancer syndromes.
###
FINANCIAL DISCLOSURE: This work was supported by Asociación Española Contra el Cancer (AECC) and Spanish Fondo de Investigaciones Sanitarias (grant numbers FISPI081298 and FIS-PI081120). The CIBER de Enfermedades Raras is an initiative of the ISCIII. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
COMPETING INTERESTS: The authors have declared that no competing interests exist.
CITATION: Martinez-Delgado B, Yanowsky K, Inglada-Perez L, Domingo S, Urioste M, et al. (2011) Genetic Anticipation Is Associated with Telomere Shortening in Hereditary Breast Cancer. PLoS Genet 7(7): e1002182. doi:10.1371/journal.pgen.1002182
Contact:
Dr. Beatriz Martinez-Delgado and Dr. Javier Benitez
Spanish National Cancer Research Centre (CNIO)
Human Genetics
Melchor Fernandez Almagro 3
Madrid 28029
SPAIN
[email protected]
[email protected]
Disclaimer
This press release refers to an upcoming article in PLoS Genetics. The release is provided by journal staff, or by the article authors and/or their institutions. Any opinions expressed in this release or article are the personal views of the journal staff and/or article contributors, and do not necessarily represent the views or policies of PLoS. PLoS expressly disclaims any and all warranties and liability in connection with the information found in the releases and articles and your use of such information.
About PLoS Genetics
[googlead tip=”lista_medie” aliniat=”stanga”]PLoS Genetics (http://www.plosgenetics.org) reflects the full breadth and interdisciplinary nature of genetics and genomics research by publishing outstanding original contributions in all areas of biology. All works published in PLoS Genetics are open access. Everything is immediately and freely available online throughout the world subject only to the condition that the original authorship and source are properly attributed. Copyright is retained by the authors. The Public Library of Science uses the Creative Commons Attribution License. [googlead tip=”lista_medie” aliniat=”centrat”]
About the Public Library of Science
The Public Library of Science (PLoS) is a non-profit organization of scientists and physicians committed to making the world’s scientific and medical literature a freely available public resource. For more information, visit http://www.plos.org.
Potential breakthrough in cancer research: a new treatment for leukemia had amazing results.







