Home Tags Posts tagged with "stem cells"
stem cells
According to Swedish scientists, stem cells can be used to heal the damage in the brain caused by Parkinson’s disease.
They said their study on rats heralded a “huge breakthrough” towards developing effective treatments.
There is no cure for the disease, but medication and brain stimulation can alleviate symptoms.
Parkinson’s disease is caused by the loss of nerve cells in the brain that produce the chemical dopamine ,which helps to control mood and movement.
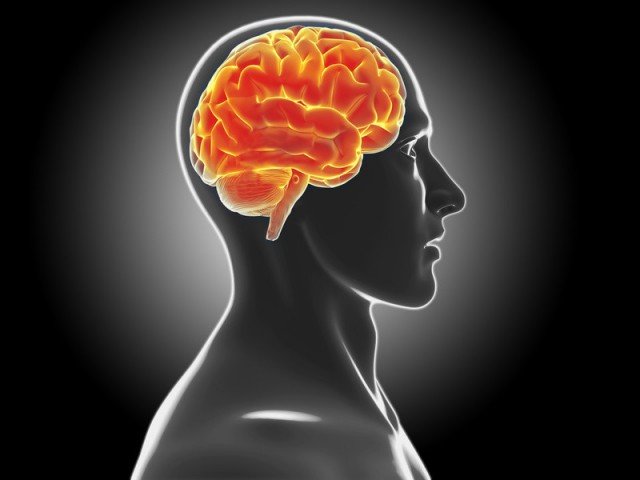
Stem cells can be used to heal brain damages caused by Parkinson’s disease
To simulate Parkinson’s, Lund University researchers killed dopamine-producing neurons on one side of the rats’ brains.
They then converted human embryonic stem cells into neurons that produced dopamine.
These were injected into the rats’ brains, and the researchers found evidence that the damage was reversed.
There have been no human clinical trials of stem-cell-derived neurons, but the researchers said they could be ready for testing by 2017.
Malin Parmar, associate professor of developmental and regenerative neurobiology, said: “It’s a huge breakthrough in the field [and] a stepping stone towards clinical trials.”
A similar method has been tried in a limited number of patients.
It involved taking brain tissue from multiple aborted foetuses to heal the brain.
Clinical trials were abandoned after mixed results, but about a third of the patients had foetal brain cells that functioned for 25 years.
Using embryonic stem cells may be preferable, as it is easier to get hold of the large numbers of cells needed for transplant by growing them in the laboratory.
It also opens up the possibility of using less ethically charged sources of stem cells, such as those made from adult tissue.
[youtube as1XnGmepyY 650]
Stem cells could be turned into killing machines to fight brain cancer, scientists from Harvard Medical School have discovered.
In experiments on mice, the stem cells were genetically engineered to produce and secrete toxins which kill brain tumors, without killing normal cells or themselves.
Researchers said the next stage was to test the procedure in humans.
A stem cell expert said this was “the future” of cancer treatment.
The study, published in the journal Stem Cells, was the work of scientists from Massachusetts General Hospital and the Harvard Stem Cell Institute.
For many years, they had been researching a stem-cell-based therapy for cancer, which would kill only tumor cells and no others.
They used genetic engineering to make stem cells that spewed out cancer-killing toxins, but, crucially, were also able to resist the effects of the poison they were producing.
They also posed no risk to normal, healthy cells.
In animal tests, the stem cells were surrounded in gel and placed at the site of the brain tumor after it had been removed.
Their cancer cells then died as they had no defense against the toxins.

Stem cells could be turned into killing machines to fight brain cancer
Dr. Khalid Shah, lead author and director of the molecular neurotherapy and imaging lab at Massachusetts General Hospital and Harvard Medical School, said the results were very positive.
“After doing all of the molecular analysis and imaging to track the inhibition of protein synthesis within brain tumors, we do see the toxins kill the cancer cells.”
He added: “Cancer-killing toxins have been used with great success in a variety of blood cancers, but they don’t work as well in solid tumors because the cancers aren’t as accessible and the toxins have a short half-life.”
But genetically engineering stem cells has changed all that, he said.
“Now, we have toxin-resistant stem cells that can make and release cancer-killing drugs.”
Dr. Khalid Shah now plans to test the technique using a number of different therapies on mice with glioblastoma, the most common brain tumor in human adults. He hopes the therapies could be used in clinical trials within the next five years.
Stem cell researchers in Japan are heralding a “major scientific discovery”, with the potential to start a new age of personalized medicine.
Scientists showed stem cells can now be made quickly just by dipping blood cells into acid.
Stem cells can transform into any tissue and are already being trialed for healing the eye, heart and brain.
The latest development, published in the journal Nature, could make the technology cheaper, faster and safer.
The human body is built of cells with a specific role – nerve cells, liver cells, muscle cells – and that role is fixed.
Stem cell researchers in Japan are heralding a “major scientific discovery”, with the potential to start a new age of personalized medicine
However, stem cells can become any other type of cell, and they have become a major field of research in medicine for their potential to regenerate the body.
Embryos are one, ethically charged, source of stem cells. Nobel Prize winning research also showed that skin cells could be “genetically reprogrammed” to become stem cells (termed induced pluripotent stem cells).
Now a study shows that shocking blood cells with acid could also trigger the transformation into stem cells – this time termed STAP (stimulus-triggered acquisition of pluripotency) cells.
Dr. Haruko Obokata, from the Riken Centre for Developmental Biology in Japan, said she was “really surprised” that cells could respond to their environment in this way.
She added: “It’s exciting to think about the new possibilities these findings offer us, not only in regenerative medicine, but cancer as well.”
The breakthrough was achieved in mouse blood cells, but research is now taking place to achieve the same results with human blood.
Frankenburger, the world’s first lab-grown burger, is to be unveiled and eaten at a news conference in London on Monday.
Scientists took cells from a cow and, at an institute in the Netherlands, turned them into strips of muscle which they combined to make a patty.
Researchers say the technology could be a sustainable way of meeting what they say is a growing demand for meat.
Critics say that eating less meat would be an easier way to tackle predicted food shortages.
Professor Mark Post of Maastricht University, the scientist behind the burger, said: “Later today we are going to present the world’s first hamburger made in a lab from cells. We are doing that because livestock production is not good for the environment, it is not going to meet demand for the world and it is not good for animals.”
But Prof. Tara Garnett, head of the Food Policy Research Network at Oxford University, said decision-makers needed to look beyond technological solutions.
“We have a situation where 1.4 billion people in the world are overweight and obese, and at the same time one billion people worldwide go to bed hungry,” she said.
“That’s just weird and unacceptable. The solutions don’t just lie with producing more food but changing the systems of supply and access and affordability so not just more food but better food gets to the people who need it.”
Stem cells are the body’s “master cells”, the templates from which specialized tissue, such as nerve or skin cells develop.
Frankenburger, the world’s first lab-grown burger, is to be unveiled and eaten at a news conference in London
Most institutes working in this area are trying to grow human tissue for transplantation, to replace worn out or diseased muscle, nerve cells or cartilage.
Mark Post wants to use similar techniques to grow muscle and fat for food.
He starts with stem cells extracted from cow muscle tissue. In the laboratory, these are cultured with nutrients and growth promoting chemicals to help them develop and multiply. Three weeks later, there are more than a million stem cells which are put into smaller dishes where they coalesce into small strips of muscle about a centimetre long and a few millimetres thick.
These strips are collected into small pellets which are frozen. When there are enough, they are defrosted and compacted into a patty just before being cooked.
The scientists have tried to make the meat – which is initially white in color – as authentic as possible. Helen Breewood, who is working with Prof. Mark Post, makes the lab-grown muscle look red by adding the naturally occurring compound myoglobin.
Currently, this is a work in progress. The burger to be revealed on Monday will be colored red with beetroot juice. The researchers have also added breadcrumbs, caramel and saffron, which will add to the taste.
At the moment, scientists can only make small pieces of meat; larger ones would require artificial circulatory systems to distribute nutrients and oxygen.
Prof. Mark Post said initial sampling suggests the burger will not taste great, but he expected it to be “good enough”.
Helen Breewood is a vegetarian because she believes meat production to be waste of resources, but says she would eat lab-grown meat.
“A lot of people consider lab-grown meat repulsive at first. But if they consider what goes into producing normal meat in a slaughter house I think they would also find that repulsive,” she said.
In a statement, animal welfare campaigners People for the Ethical Treatment of Animals (PETA) said: “[Lab-grown meat] will spell the end of lorries full of cows and chickens, abattoirs and factory farming. It will reduce carbon emissions, conserve water and make the food supply safer.”
The latest United Nations Food and Agriculture Organization report on the future of agriculture indicates that most of the predicted growth in demand for meat from China and Brazil has already happened and many Indians are wedded to their largely vegetarian diets for cultural and culinary reasons.
Lab grown meat might turn out to be a technological solution in search of a problem.
[youtube 27PO_nu3BEs]
[youtube VikkBQRtbdk]
Frankenburger is the world’s first test-tube burger and is being served in London.
The burger is made from meat grown in a laboratory, rather than cattle raised in pastures.
And its developers hope it will show how the soaring global demand for protein can be met without the need for vast herds of cattle.
The 5oz Frankenburger, which cost £250,000 ($400,000) to produce, is made from 3,000 tiny strips of meat grown from the stem cells of a cow.
The raw meat is said to be grey with a slippery texture similar to squid or scallop.
It has been created by Professor Mark Post, from the University of Maastricht in Holland.
His research has been funded by an anonymous businessman – who may be the first to try the burger.
Prof. Mark Post told The Independent on Sunday: “Right now, we are using 70 per cent of all our agricultural capacity to grow meat through livestock. You are going to need alternatives. If we don’t do anything meat will become a luxury food and will become very expensive.”
A four-step technique is used to turn stem cells from animal flesh into a burger.
First, the stem cells are stripped from the cow’s muscle.
Frankenburger is the world’s first test-tube burger and is being served in London
Next, they are incubated in a nutrient broth until they multiply many times over, creating a sticky tissue with the consistency of an undercooked egg.
This “wasted muscle” is then bulked up through the laboratory equivalent of exercise – it is anchored to Velcro and stretched.
Finally, 3,000 strips of the lab-grown meat are minced, and, along with 200 pieces of lab-grown animal fat, formed into a burger.
The process is still lengthy, as well as expensive, but it could take just six weeks from stem cell to supermarket shelf.
Mark Post’s work is funded by the Dutch government, as well as an anonymous donation of 300,000 euros – but it remains to be seen, however, whether the pioneering development will find favor with a public that likes to think of its chops, steaks and sausages as having their roots in nature, rather than in test-tubes.
He first attempts involved mouse burgers. He then tried to grow pork in a dish, producing strips with the rubbery texture of squid or scallops, before settling on beef.
Mark Post’s burger consists of about 20,000 thin strips of cultured muscle tissue.
The cell-grown burger is produced with materials – including fetal calf serum, which used to grow the cells – that will eventually be replaced by materials not originating from animals, the New York Times reported.
In-vitro meat or cultured meat is an animal flesh product that has never been part of a complete, living animal, and is quite different from imitation meat or meat substitutes, which are vegetarian foods made from vegetable proteins like soy.
Scientists say that it is possible the meat will be sold to the public within ten years.
It also reduces the amount of feed, water and fuel needed to produce beef.
Every kilo of meat requires 10 kilos of plant feed and oil, but cultured meat would only need two.
According to the Food and Agricultural Organization we will be eating twice as much meat as we do now by 2050.
Prof. Mark Post added: “It comes down to the fact that animals are very inefficient at converting vegetable protein into animal protein. This helps drive up the cost of meat.”
It is thought that the new form of meat could be acceptable to vegetarians, and animal rights organizations have already given their approval.
PETA spokesman Ben Williamson said: “We do support lab-grown meat if it means fewer animals are eaten. Anything that reduces the suffering of animals would be welcome.”
PETA also runs a competition offering a $1 million prize for the person who invents an artificial form of chicken meat.
Oxford university scientists said in 2011 that cultured beef would need 45% less energy that natural beef.
They added that it would require 99% less land than regular livestock and produce between 78 and 95% less greenhouse gas.
But, the difficulty may be persuading the public to eat an artificial product.
Prof. Mark Post said that it is possible to add fatty tissue and nutrients to it, changing the taste and making it more palatable for the public.
[youtube 6D5Cv3tDhmA]
A Chinese research team has grown rudimentary teeth out of the most unlikely of sources, human urine.
The results, published in Cell Regeneration Journal, showed that urine could be used as a source of stem cells that in turn could be grown into tiny tooth-like structures.
The scientists hope the technique could be developed into a way of replacing lost teeth.
Other stem cell researchers caution that that goal faces many challenges.
Teams of researchers around the world are looking for ways of growing new teeth to replace those lost with age and poor dental hygiene.
Stem cells – the master cells which can grow into any type of tissue – are a popular area of research.

The Chinese research team has grown rudimentary teeth out of the most unlikely of sources, human urine
The group at the Guangzhou Institutes of Biomedicine and Health used urine as the starting point.
Cells which are normally passed from the body, such as those from the lining of the body’s waterworks, are harvested in the laboratory. These collected cells are then coaxed into becoming stem cells.
A mix of these cells and other material from a mouse was implanted into the animals.
The researchers said that after three weeks the bundle of cells started to resemble a tooth: “The tooth-like structure contained dental pulp, dentin, enamel space and enamel organ.”
However, the “teeth” were not as hard as natural teeth.
This piece of research is not immediately going to lead to new options for the dentist, but the researchers say it could lead to further studies towards “the final dream of total regeneration of human teeth for clinical therapy”.
John Gurdon from the UK and Shinya Yamanaka from Japan, two pioneers of stem cell research, have shared the Nobel Prize for medicine or physiology.
John Gurdon and Shinya Yamanaka were awarded the prize for transforming specialized cells into stem cells, which can become any other type of cell in the body.
Prof. John Gurdon used a gut sample to clone frogs and Prof. Shinya Yamanaka altered genes to reprogramme cells.
The Nobel committee said they had “revolutionized” science.

Prof. John Gurdon used a gut sample to clone frogs and Prof. Shinya Yamanaka altered genes to reprogramme cells
In 1962, John Gurdon took the genetic information from a cell in the intestines of a frog and placed it inside a frog egg, which developed into a normal tadpole.
Shinya Yamanaka showed that specialized mouse cells could be reprogrammed to become stem cells by introducing four genes. The resulting stem cells could then be converted to other types of cell.
The Nobel committee said the discovery had “revolutionized our understanding of how cells and organisms develop”.
Stem cells made from skin have become “grandparents” after generations of life were created in experiments by scientists in Japan.
The cells were used to create eggs, which were fertilized to produce baby mice. These later had their own babies.
If the technique could be adapted for people, it could help infertile couples have children and even allow women to overcome the menopause.
But experts say many scientific and ethical hurdles must be overcome.
Stem cells are able to become any other type of cell in the body from blood to bone, nerves to skin.
Last year the team at Kyoto University managed to make viable sperm from stem cells. Now they have performed a similar feat with eggs.
They used stem cells from two sources: those collected from an embryo and skin-like cells which were reprogrammed into becoming stem cells.
The first step, reported in the journal Science, was to turn the stem cells into early versions of eggs.
A “reconstituted ovary” was then built by surrounding the early eggs with other types of supporting cells which are normally found in an ovary. This was transplanted into female mice.
Surrounding the eggs in this environment helped them to mature.
IVF techniques were used to collect the eggs, fertilize them with sperm from a male mouse and implant the fertilized egg into a surrogate mother.
Dr. Katsuhiko Hayashi, from Kyoto University, said: “They develop to be healthy and fertile offspring.”
Those babies then had babies of their own, whose “grandmother” was a cell in a laboratory dish.
The ultimate aim of the research is to help infertile couples have children. If the same methods could be used in people then cells in skin could be turned into an egg. Any resulting child would be genetically related to the mother.
However, Dr. Katsuhiko Hayashi said that was still a distant prospect: “I must say that it is impossible to adapt immediately this system to human stem cells, due to a number of not only scientific reasons, but also ethical reasons.”
He said that the level of understanding of human egg development was still too limited. There would also be questions about the long-term consequences on the health of any resulting child.
Prof. Robert Norman, from the University of Adelaide, said: “For many infertile couples, finding they have no sperm or eggs is a devastating blow.
“This paper offers light to those who want a child, who is genetically related to them, by using personalized stem cells to create eggs that can produce an offspring that appears to be healthy.
“It also offers the potential for women to have their own children well past menopause raising even more ethical issues.
“Application to humans is still a long way off, but for the first time the goal appears to be in sight.”
A jab that allows damaged hearts to heal themselves and could be given by paramedics in the back of ambulances is being developed at a British university.
Scientists at Imperial College London hope that giving heart attack victims an injection of stem cells will trick the organ into repairing itself, saving lives and greatly cutting the odds of further ill health.
Crucially, and unlike other techniques being tested on patients in the UK, the cells they plan to use are from a person’s own heart, an innovation they believe increases the odds of the treatment being a success.
They are close to applying for permission to test the jab on patients.
If trials on heart attack survivors are successful, the injection could eventually be given by paramedics just minutes after a heart attack and before patients even reach hospital.

A jab that allows damaged hearts to heal themselves and could be given by paramedics in the back of ambulances is being developed at Imperial College London
The jab is one of several treatments being researched by the British Heart Foundation as part of its multi-million-pound Mending Broken Hearts project to improve the care of heart attack patients.
The aim is to cut the odds of heart failure, in which the heart, weakened by one, or a series of heart attacks, struggles to pump blood around the body.
In the most severe cases, the lungs ‘drown’ in fluid.
Treatments range from drugs to transplants but with 40% of those affected dying within a year of diagnosis, heart failure has a worse survival rate than many cancers.
Doctors and scientists around the world are trying to use stem cells – “blank” cells able to turn into various types of tissue – to shore up ailing hearts.
But most have focused on cells taken from bone marrow and improvements have been slight.
The Imperial team believes that stem cells from the heart will be much more successful.
These cells are extremely rare, with just 300 per million normal heart cells, meaning the lack the power needed to repair the damage wrought by a heart attack.
But the scientists have found a way of extracting them from a patient’s own heart, and growing them in huge numbers the laboratory, before injecting them back into the hear
Once there, they patch up the ailing tissue, with tests on mice showing stem cells taken from the animals’ hearts trigger the growth of new tissue and blood vessels.
The human version of the jab has also been tested on pigs and the researchers are one to two years away from applying for permission to test the treatment on patients.
With it taking three to four months to grow enough cells for each jab, the first patients will be treated several months after a heart attack.
But in time, it may be possible to create a one-size-fits-all jab, allowing almost immediate treatment, said researcher Professor Michael Schneider.
Esther Rantzen, who is backing the Mending Broken Hearts appeal and whose late husband, the documentary maker, Desmond Wilcox battled heart disease for years, said: “If hearts learn to heal themselves, then people who are bed-bound, who are imprisoned in their own homes, who can’t walk upstairs, who can’t involve themselves in any physical activity could be restored to health and their family life greatly improved.”
Prof. Michael Schneider is also trying to find ways of stopping cells from dying during a heart attack.
Other work being funded by the BHF includes research into a pill that could be given in advance to those at high risk of heart attacks and patches of cells that could patch up the heart.
Professor Peter Weissberg, the charity’s medical director, said that despite advances in cardiac medicine, a good treatment for severe heart failure has remained elusive.
“Although we have been able to prevent heart attacks and treat people with heart attacks when they occur, we haven’t been able to stop the damage that occurs when a heart attack takes place.
“The reason we are making such a noise about it now is that science has progressed to a point where it looks biologically feasible that we might be able to create new heart cells to repair the heart.
“Ten years ago, that would have been science function.”
British researchers say they have taken a huge step forward in treating deafness after stem cells were used to restore hearing in animals for the first time.
Hearing partially improved when nerves in the ear, which pass sounds into the brain, were rebuilt in gerbils – a UK study in the journal Nature reports.
Getting the same improvement in people would be a shift from being unable to hear traffic to hearing a conversation.
However, treating humans is still a distant prospect.

British researchers say they have taken a huge step forward in treating deafness after stem cells were used to restore hearing in animals for the first time
If you want to listen to the radio or have a chat with a friend your ear has to convert sound waves in the air into electrical signals which the brain will understand.
This happens deep inside the inner ear where vibrations move tiny hairs and this movement creates an electrical signal.
However, in about one in 10 people with profound hearing loss, nerve cells which should pick up the signal are damaged. It is like dropping the baton after the first leg of a relay race.
The aim of researchers at the University of Sheffield was to replace those baton-dropping nerve cells, called spiral ganglion neurons, with new ones.
They used stem cells from a human embryo, which are capable of becoming any other type of cell in the human body from nerve to skin, muscle to kidney.
A chemical soup was added to the stem cells that converted them into cells similar to the spiral ganglion neurons. These were then delicately injected into the inner ears of 18 deaf gerbils.
Over 10 weeks the gerbils’ hearing improved. On average 45% of their hearing range was restored by the end of the study.
Dr. Marcelo Rivolta said: “It would mean going from being so deaf that you wouldn’t be able to hear a lorry or truck in the street to the point where you would be able to hear a conversation.
“It is not a complete cure, they will not be able to hear a whisper, but they would certainly be able to maintain a conversation in a room.”
About a third of the gerbils responded really well to treatment with some regaining up to 90% of their hearing, while just under a third barely responded at all.
Gerbils were used as they are able to hear a similar range of sounds to people, unlike mice which hear higher-pitched sounds.
The researchers detected the improvement in hearing by measuring brainwaves. The gerbils were also tested for only 10 weeks. If this became a treatment in humans then the effect would need to be shown over a much longer term.
There are also questions around the safety and ethics of stem cell treatments which would need to be addressed.
Researchers have discovered the cells in tumors that seem to be responsible for their re-growth.
Three separate studies on mice appear to have confirmed the view that the growth of tumors is driven by so-called cancer stem cells.
The researchers claim to have resolved one of the biggest controversies in cancer research and say their work marks a “paradigm shift” in the field.
The studies have been published in the journals, Nature and Science.
Doctors often successfully reduce the size of tumors through various therapies, but often patients suffer a relapse and the tumor re-grows.
Three separate studies on mice appear to have confirmed the view that the growth of tumors is driven by so-called cancer stem cells
Some researchers believe that this happens because therapies fail to eradicate a small proportion of cells that drive tumor growth known as cancer stem cells. They believe that these are the cells that should be targeted to eliminate the tumor forever.
Evidence for the existence of cancer stem cells has been weak. But now three separate groups of researchers working independently have found direct evidence of cancer stem cells driving tumor growth in brain, gut and skin cancers.
The suggestion is that the same may be true of all cancers which produce solid tumors.
According to Prof. Cedric Blanpain of the Free University of Brussels, who led one of the studies, the results could pave the way for a new approach to treating many cancers.
“If these cells are indeed the cells that fuel tumour growth then maybe you can target these cells,” he said.
But that may be easier said than done. The newly-identified cancer stem cells are very similar to healthy stem cells responsible for growing and renewing tissue in the body. Any therapy to target cancer stem cells may also destroy healthy tissues. A priority for researchers will be to see if there are important differences between normal and cancer stem cells so that therapies can distinguish between them.
But according to Prof. Hugo Snippert of the University Medical Centre in Utrecht, who led the study into intestinal tumors, the confirmation that these cells exist is an important step in future cancer research.
“Many argued that these cells did not exist. But we have shown for the first time there is such a thing as a cancer stem cell and that tumors are maintained by them,” he said.
Prof. Luis Parada of the University of Texas, who led research that identified stem cells in brain tumors in mice, said he believed there would now be a new approach to developing new treatments for solid tumor cancers.
“Cancer stem cells change the paradigm. The goal of shrinking tumors may well turn out to be less important than targeting the cancer cells in that tumor.”
Early results from a US trial suggest it may be possible to use “stem cell shielding” to protect the body from the damaging effects of chemotherapy.
Chemotherapy drugs try to kill rapidly dividing cancer cells, but they can also affect other healthy tissues such as bone marrow.
The study, published in Science Translational Medicine, used genetically modified stem cells to protect the bone marrow.
The body constantly churns out new blood cells in the hollow spaces inside bone. However, bone marrow is incredibly susceptible to chemotherapy.
The treatment results in fewer white blood cells being produced, which increase the risk of infection, and fewer red blood cells, which leads to shortness of breath and tiredness.

Early results from a US trial suggest it may be possible to use "stem cell shielding" to protect the body from the damaging effects of chemotherapy
Researchers at the Fred Hutchinson Cancer Research Center, in Seattle, said these effects were “a major barrier” to using chemotherapy and often meant the treatment had to be stopped, delayed or reduced.
They have tried to protect the bone marrow in three patients with a type of brain cancer, glioblastoma.
One of the researchers, Dr. Jennifer Adair, said: “This therapy is analogous to firing at both tumor cells and bone marrow cells, but giving the bone marrow cells protective shields while the tumor cells are unshielded.”
Bone marrow was taken from the patients and stem cells, which produce blood, were isolated. A virus was then used to infect the cells with a gene which protected the cells against a chemotherapy drug. The cells were then put back into the patient.
The lead author of the report, Prof. Hans-Peter Kiem, said: “We found that patients were able to tolerate the chemotherapy better, and without negative side effects, after transplantation of the gene-modified stem cells than patients in previous studies who received the same type of chemotherapy without a transplant of gene-modified stem cells.”
The researchers said the three patients had all lived longer than the average survival time of 12 months for the cancer. They said one patient was still alive 34 months after treatment.











