Home Tags Posts tagged with "chromosome"
chromosome
Experts found that men can inherit heart disease from their father after they have tracked the condition to the Y chromosome that dads pass to sons.
By studying the DNA of over 3,000 men, scientists found a particular version of the sex chromosome increases the risk of coronary artery disease by 50%.
The risk it confers is in addition to other heart risk factors like cholesterol, The Lancet reports.
Experts already know that men develop heart disease a decade earlier than women, on average. By the age of 40, the lifetime risk of heart disease is one in two for men and one in three for women.
Lifestyle factors like smoking and blood pressure are important contributors. This latest work suggests the male Y chromosome can also play a role in coronary artery disease.
Dr. Maciej Tomaszewski, from the University of Leicester in UK, and colleagues studied 3,233 biologically unrelated British men who were already enrolled in other medical studies investigating heart disease risk.
When they carried out genetic tests on the men they found that 90% possessed one of two common versions of Y chromosome – named haplogroup I and haplogroup R1b1b2.
And the risk of coronary artery disease among the men carrying the haplogroup I version was 50% higher than in other men.
The scientists say they now need to pinpoint precisely which genes on the Y chromosome are responsible.
But they believe they already know how they exert their effect – by upsetting a man’s immune system.
Dr. Maciej Tomaszewski, a clinical senior lecturer at the University’s Department of Cardiovascular Sciences, said: “We are very excited about these findings as they put the Y chromosome on the map of genetic susceptibility to coronary artery disease.
“Doctors usually associated the Y chromosome with maleness and fertility but this shows it is also implicated in heart disease.”
Experts found that men can inherit heart disease from their father after they have tracked the condition to the Y chromosome that dads pass to sons
He said, ultimately, the discovery could lead to new ways to treat and prevent heart disease in men, as well as a genetic test to spot those at greatest risk.
In the meantime, Dr. Maciej Tomaszewski said men should focus on risk factors that they already have the power to modify themselves, such as getting enough exercise and eating a healthy diet to keep their blood pressure and cholesterol down.
Dr. Hélène Wilson of the British Heart Foundation, which part-funded the work, said: “Coronary heart disease is the cause of heart attacks, which claim the lives of around 50,000 UK men every year.
“Lifestyle choices such as poor diet and smoking are major causes, but inherited factors carried in DNA are also part of the picture. The next step is to identify specifically which genes are responsible and how they might increase heart attack risk.”
Scientists say life expectancy is written into our DNA and could be seen from the day we are born.
They have found a way to predict how long someone will live – by measuring their genes as a baby
It all depends on the length of the telomeres, which are described as “acting like the plastic ends on shoelaces” to protect chromosomes from wear and tear.
Telomeres are being studied extensively – and are thought to hold the key to ageing.
The longer your telomeres, the longer you will live – dependent, of course, on not dying accidentally, from disease or from lifestyle factors.
It was known they could be shortened by life choices, including smoking and stress. But this is the first indication that our lifespan might be predetermined from birth.
In the future, tests may allow people to know their expected lifespan from a very early age – if they want to.
Professor Pat Monaghan, who led the Glasgow University study, said: “The results of this research show that what happens in our bodies in early life is very important.
“It is not understood why there are variations of telomere length but if you had a choice, you would want to be born with longer telomeres.
“If you were to test this, I don’t think anyone would want to know – it would just make you miserable. But it must be remembered that how you live has a big effect. This isn’t quite a case of nature overtaking nurture.”
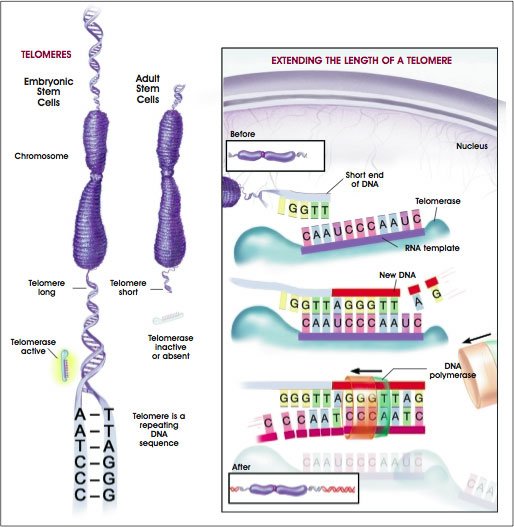
Telomeres are important because they stop DNA from unraveling, but they begin shortening from the moment we are conceived
The study – which used zebra finches, one of Australia’s most common bird species – is the first to measure telomere lengths at regular intervals through an entire life. With people, it is usually only the elderly who are studied because of the timescales involved.
Blood cell samples were taken from 99 finches, starting when they were 25 days old.
The results exceeded even the researchers’ expectations. The birds with the shortest telomeres did tend to die first – from as early as seven months after the start of the trial.
But one bird in the group with the longest telomeres survived to almost nine years old.
Professor Monaghan said: “These birds were dying of natural causes. There were no predators, no diseases and no accidental deaths. This was showing their capacity for long life.”
The results hold huge implications for humans, whose telomeres work in the same way.
Telomeres are important because they stop DNA from unraveling, but they begin shortening from the moment we are conceived.
The longer they are, the better for an individual because when they get too short, they stop working.
DNA is then no longer protected and errors begin to creep in when cells divide. When this happens – usually in middle age – the skin begins to sag and the immune system becomes less efficient. Faulty cells also lead to a growing risk of conditions such as diabetes and heart disease.
The university’s institute of biodiversity, animal health and comparative medicine has published its groundbreaking research in the Proceedings of the National Academy of Sciences USA.
In the next stage of their research, the Glasgow scientists will look at what causes telomeres to shorten – including inherited and environmental factors – to make it possible to predict life expectancy more accurately.
[googlead tip=”patrat_mare” aliniat=”stanga”]
Progressive telomere shortening characterizes familial breast cancer patients
Telomere chromosome
Telomeres, the complex structures that protect the end of chromosomes, of peripheral blood cells are significantly shorter in patients with familial breast cancer than in the general population. Results of the study carried out by the Human Genetics Group of the Spanish National Cancer Research Centre (CNIO), led by Javier Benitez, to be published in open-access journal PLoS Genetics on July 28th, reflect that familial, but not sporadic, breast cancer cases are characterized by shorter telomeres. Importantly, they also provide evidence for telomere shortening as a mechanism of genetic anticipation, the successively earlier onset of cancer down generations.
Mutations in two DNA repair genes, BRCA1 and BRCA2, characterize some, but not all, instances of hereditary breast cancer. Non-BRCA1/2 breast cancer families are heterogeneous, suggesting the existence of other genes conferring susceptibility. The group has investigated the role of telomere length in hereditary breast cancer based on previous information suggesting, first, that short telomeres and subsequent genomic instability contribute to malignant transformation; second, that genetic anticipation occurs in breast cancer families and, third, that telomere shortening is associated with anticipation in other genetic diseases. [googlead tip=”vertical_mare” aliniat=”stanga”]
By analyzing telomere length differences between mothers and daughters from breast cancer families, the authors demonstrated that genetic anticipation is associated with a decrease in telomere length in affected daughters relative to their mothers.
The results allowed the authors not only to conclude that women carrying BRCA1/2 mutation have chromosomes with short telomeres, but also to describe for the first time that genetic anticipation in breast cancer could be explained by telomere shortening. In addition, the study expands the field of research concerning genetic predisposition to breast cancer to include genes involved in telomere maintenance. The significance of generational changes in telomere length has interesting potential clinical applications in the management of familial breast cancer, and could be extended to other hereditary cancer syndromes.
###
FINANCIAL DISCLOSURE: This work was supported by Asociación Española Contra el Cancer (AECC) and Spanish Fondo de Investigaciones Sanitarias (grant numbers FISPI081298 and FIS-PI081120). The CIBER de Enfermedades Raras is an initiative of the ISCIII. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
COMPETING INTERESTS: The authors have declared that no competing interests exist.
CITATION: Martinez-Delgado B, Yanowsky K, Inglada-Perez L, Domingo S, Urioste M, et al. (2011) Genetic Anticipation Is Associated with Telomere Shortening in Hereditary Breast Cancer. PLoS Genet 7(7): e1002182. doi:10.1371/journal.pgen.1002182
Contact:
Dr. Beatriz Martinez-Delgado and Dr. Javier Benitez
Spanish National Cancer Research Centre (CNIO)
Human Genetics
Melchor Fernandez Almagro 3
Madrid 28029
SPAIN
[email protected]
[email protected]
Disclaimer
This press release refers to an upcoming article in PLoS Genetics. The release is provided by journal staff, or by the article authors and/or their institutions. Any opinions expressed in this release or article are the personal views of the journal staff and/or article contributors, and do not necessarily represent the views or policies of PLoS. PLoS expressly disclaims any and all warranties and liability in connection with the information found in the releases and articles and your use of such information.
About PLoS Genetics
[googlead tip=”lista_medie” aliniat=”stanga”]PLoS Genetics (http://www.plosgenetics.org) reflects the full breadth and interdisciplinary nature of genetics and genomics research by publishing outstanding original contributions in all areas of biology. All works published in PLoS Genetics are open access. Everything is immediately and freely available online throughout the world subject only to the condition that the original authorship and source are properly attributed. Copyright is retained by the authors. The Public Library of Science uses the Creative Commons Attribution License. [googlead tip=”lista_medie” aliniat=”centrat”]
About the Public Library of Science
The Public Library of Science (PLoS) is a non-profit organization of scientists and physicians committed to making the world’s scientific and medical literature a freely available public resource. For more information, visit http://www.plos.org.
Potential breakthrough in cancer research: a new treatment for leukemia had amazing results.



